- Research of Doxorubicin began in the 1950's after analyzing Streptomycin peucetius.
- In 1967, cardiotoxic effects of the drug were recognized.
- In the early to mid 1990's, a liposomal version of Doxorubicin was developed. This new version was trademarked DOXIL.
- In 1996, the FDA formally approved DOXIL.
- In January, 2015, the FDA approved a new manufacturer of DOXIL.
History in Detail
Research on Doxorubicin began in the 1950's when a Italian research company called Farmilatlia Research Laboratories began a organized effort to find an anticancer compound derived from soil based microbes. They isolated the original drug from a strain of Streptomyces peucetius. At the same time, a group of French researchers discovered the drug. As such, the drug was named daunorubicin combining the name Dauni (a preRoman tribe that occupied the area where the drug was synthesized) and rubis (French for ruby, which described the color). Clinical trials began in the early 1960's and the drug was proven successful at treating leukemia and lymphoma. However, by 1967, the cardiotoxic effects of Daunorubicin were recognized.
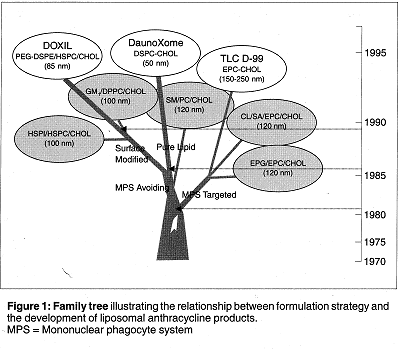
Genetic alterations were made to the Streptomyces to produce a similar, but different, antibiotic named Adriamycin; this name was later changed to Doxorubicin. This modified form of the drug was more effective in tumor treatment than Daunorubicin, but still had significant cardiotoxic effects. These two compounds became the prototype anthracyclines. As time went on, liposomal forms of the drug were developed. In addition, methods for mononuclear phagocyte avoiding systems and modification of surface proteins were developed. This allowed for specific, safer targeting of the drug.
Doxorubicin was left on the proverbial shelf because of its cardiotoxicity until the 1990's. In the early 1990's, Doxil was developed. Doxil is a liposomal drug delivery system for Doxorubicin, which alleviated the cardiotoxic symptoms. In 1996, the FDA formally approved DOXIL. Since then, Doxil has been modified and the liposomes have been altered to allow for a greater affinity for cancerous cells.
In January of 2015, the FDA approved a new manufacturer of DOXIL, which will hopefully lead to a reduced production cost.
Genetic alterations were made to the Streptomyces to produce a similar, but different, antibiotic named Adriamycin; this name was later changed to Doxorubicin. This modified form of the drug was more effective in tumor treatment than Daunorubicin, but still had significant cardiotoxic effects. These two compounds became the prototype anthracyclines. As time went on, liposomal forms of the drug were developed. In addition, methods for mononuclear phagocyte avoiding systems and modification of surface proteins were developed. This allowed for specific, safer targeting of the drug.
Doxorubicin was left on the proverbial shelf because of its cardiotoxicity until the 1990's. In the early 1990's, Doxil was developed. Doxil is a liposomal drug delivery system for Doxorubicin, which alleviated the cardiotoxic symptoms. In 1996, the FDA formally approved DOXIL. Since then, Doxil has been modified and the liposomes have been altered to allow for a greater affinity for cancerous cells.
In January of 2015, the FDA approved a new manufacturer of DOXIL, which will hopefully lead to a reduced production cost.
http://imaging.ubmmedica.com/cancernetwork/journals/oncology/images/pld02f1.gif
Tahover, E., Patil, Y., and Gabison A. 2014. Emerging delivery systems to reduce doxorubicin cardiotoxicity and improve therapeutic index: focus on liposomes. Anti-Cancer Drugs. Vol 26, No. 3: 241-258.